
That was a big puzzle before the discovery of the quantum laws that describe small things. If you were to imagine that the electrons were little classical particles in orbit, they would radiate electromagnetic waves, losing energy, and the atom would collapse in about a billionth of a second. If the orbital velocity were too small (say zero, for example), the planet would fall into the Sun. That's not a possibility for planetary motion. That's true of the electrons in hydrogen, helium, and some of the electrons in other elements. Then there is some indefinite velocity, but it's radial, i.e. The electrons in atoms are not in particular places and do not have particular orbital velocities, except when the orbital velocity is exactly zero. For example, a planet circling the Sun is at a particular place and has a particular orbital velocity. However, at least as much would be false. It's true that students could do that, and some of what it would convey about atoms would be true. You can click on any element to find information about it, and the atomic numbers are listed on the table itself, so they're easy to find.Īs for what to build your atom out of, be creative! So far, scientists have discovered elements with atomic numbers of even more than 100! For more information on different elements, check out this. Some other elements are Helium (2), Lithium (3), Beryllium (4), Boron (5), Carbon (6), Nitrogen (7), and Oxygen (8). For example, Hydrogen has one proton so its atomic number is 1. The number of protons that an atom has is called its atomic number.

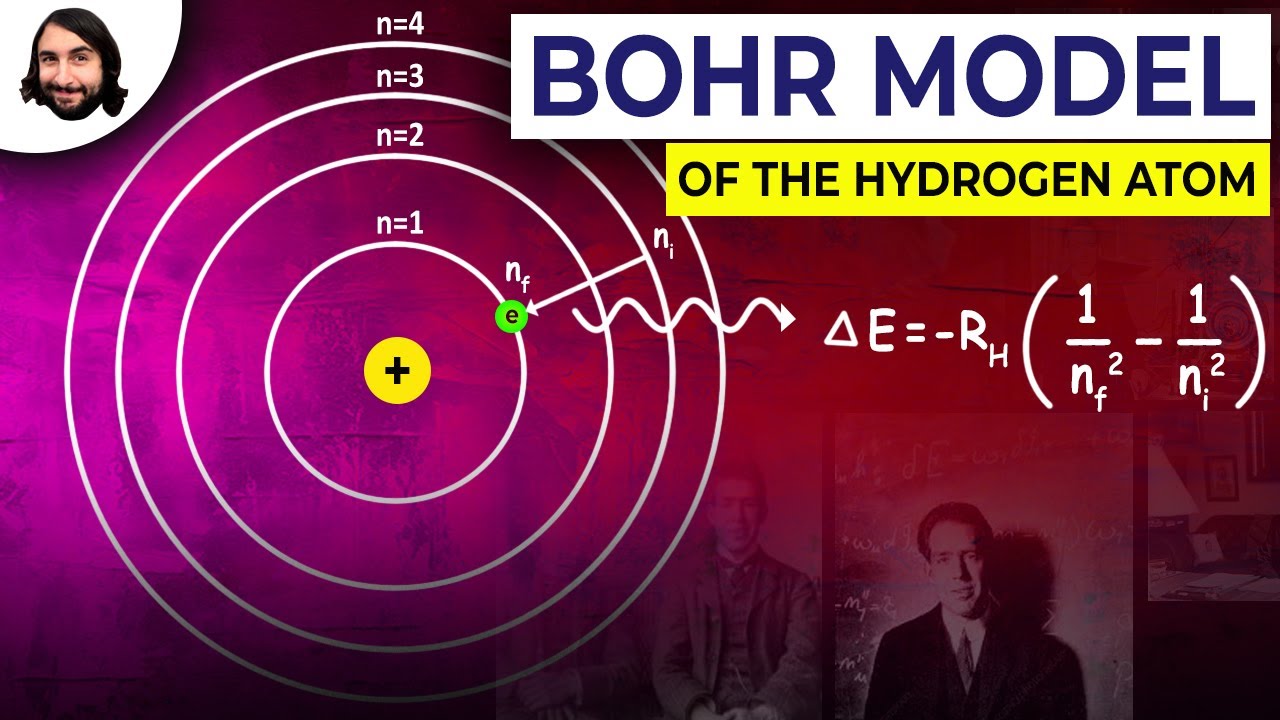
A normal gold atom, for example, has 79 protons and 118 neutrons.Īn interesting fact is that although the "cloud" of electrons that surround the nucleus is much much much bigger than the nucleus itself, most of the mass of the atom (more than 99%) is due to the nucleus. Atoms also tend to have similar numbers of neutrons and protons, though the trend is for heavier atoms to have have more neutrons than protons. If a neutral atom loses or gains some electrons it is called an ion. Neutral atoms have the same number of protons and electrons. You can use different colors to show which are neutrons, protons, or electrons. Neutrons and protons are about the same size, and electrons are much much much smaller. The atom is held together by electrostatic forces between the positive nucleus and negative surroundings.Cool project! The basic structure of an atom is that it has little things called neutrons and protons that are stuck together in a ball (called a nucleus) in the middle, with electrons in a bigger fuzzy ball around that. In the model, electrons orbit the nucleus in atomic shells. He described it as a positively charged nucleus, comprised of protons and neutrons, surrounded by a negatively charged electron cloud. Niels Bohr introduced the atomic Hydrogen model in 1913.

 RSS Feed
RSS Feed
